![PDF] Photocatalytic Barbier reaction – visible-light induced allylation and benzylation of aldehydes and ketones† †Electronic supplementary information (ESI) available. See DOI: 10.1039/c8sc02038h | Semantic Scholar PDF] Photocatalytic Barbier reaction – visible-light induced allylation and benzylation of aldehydes and ketones† †Electronic supplementary information (ESI) available. See DOI: 10.1039/c8sc02038h | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4e47c016d4cbb1df10c98e9abd9c1a88b9bc05b4/5-Figure1-1.png)
PDF] Photocatalytic Barbier reaction – visible-light induced allylation and benzylation of aldehydes and ketones† †Electronic supplementary information (ESI) available. See DOI: 10.1039/c8sc02038h | Semantic Scholar
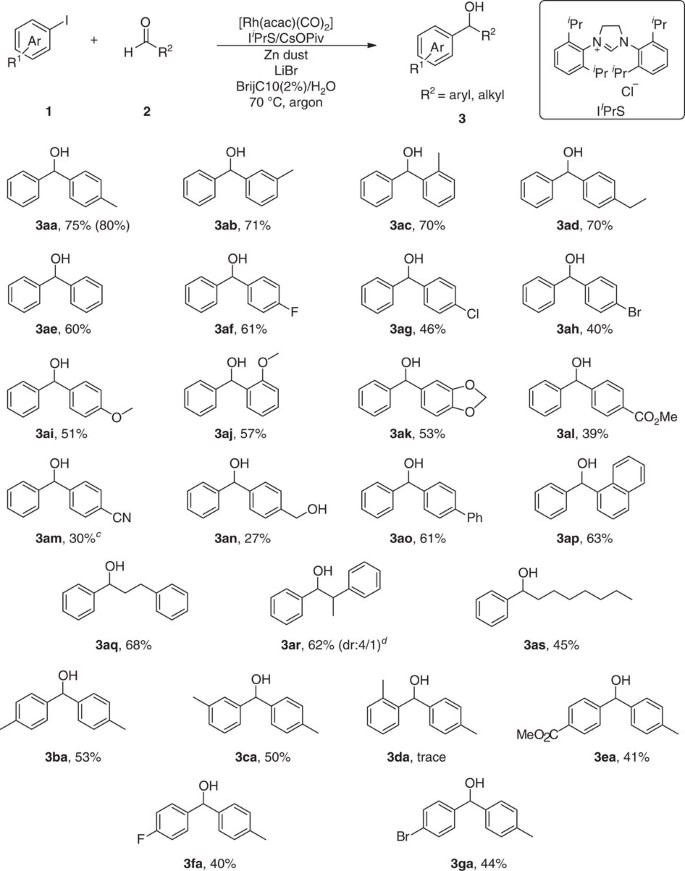
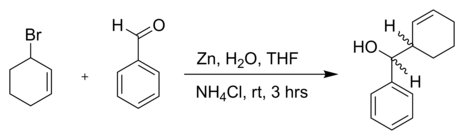
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications

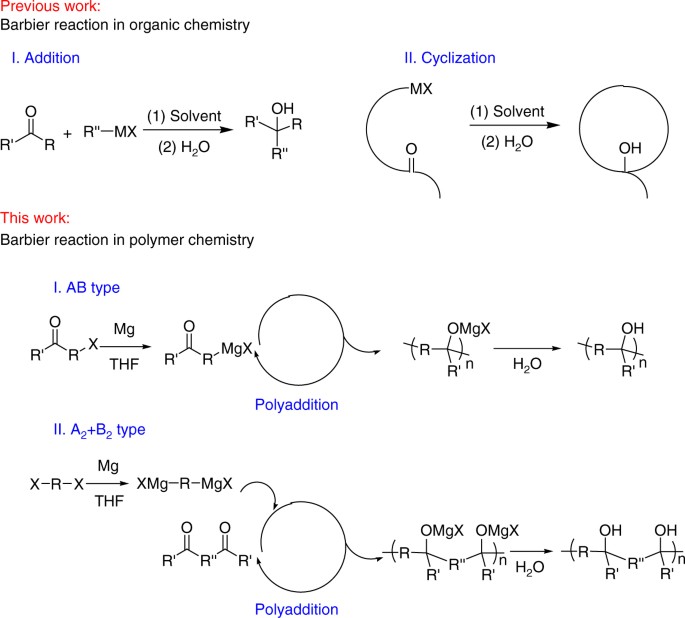
Scheme 2. Different products obtained under Barbier reaction conditions... | Download Scientific Diagram

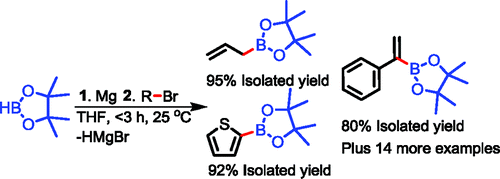
Hydride as a Leaving Group in the Reaction of Pinacolborane with Halides under Ambient Grignard and Barbier Conditions. One-Pot Synthesis of Alkyl, Aryl, Heteroaryl, Vinyl, and Allyl Pinacolboronic Esters | The Journal

The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications

Ultrasound promoted Barbier reactions and Csp3–Csp2 Stille coupling for the synthesis of diarylmethanes and substituted benzophenones - ScienceDirect
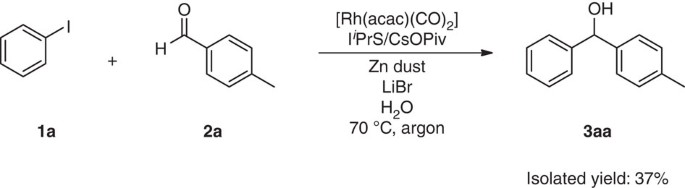
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications
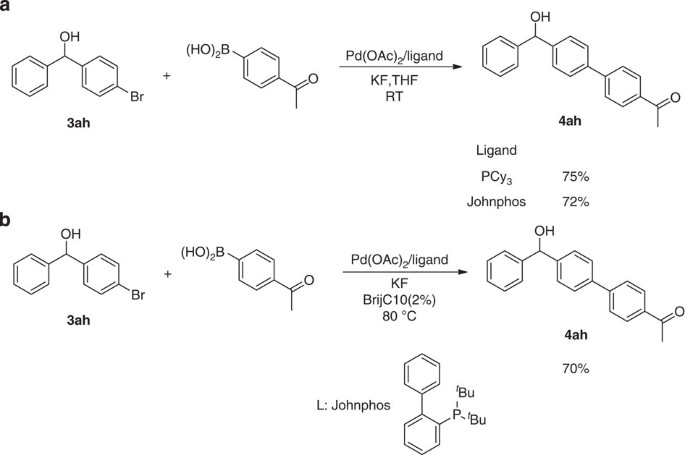
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications