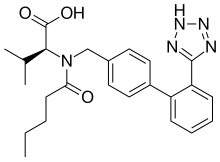
EMA recalls heart medicine valsartan made by Chinese firm over potential cancer-causing residue | Fierce Pharma

Teva Pharmaceuticals USA Issues Voluntary Nationwide Recall of all Amlodipine/Valsartan Combination Tablets and Amlodipine/Valsartan/Hydrochlorothiazide Combination Tablets that are Within Expiry | FDA

Novartis v. Teva: Patent rivalries meet again, this time on a non-negligible injunction. - The IPKat




/cloudfront-us-east-1.images.arcpublishing.com/gray/UVAUY3N3VBKKPJZ24MSVDZL62I.png)
/cloudfront-us-east-1.images.arcpublishing.com/gray/AJPIXHID3RKCJMT7TUFXT7WVEU.jpg)